Curriculum links:
- Physics and Chemistry – Density and viscosity
Learning objectives:
- Investigate the density of various liquids and solids
- Why some liquids mix, and others don’t
- Students will learn to build a colourful tower of stacked liquids with different densities.
Materials:
- Clear container, the taller and narrower the better
- Lamp oil
- Rubbing alcohol
- Vegetable oil
- Water
- Washing-up liquid
- Milk
- Maple syrup
- Corn syrup
- Honey
- Ping pong ball
- Plastic bottle cap
- Plastic bead
- Grape tomato
- Dice
- Popcorn kernel
- Metal nut or bolt
Safety:
- Adult supervision required
Instructions:
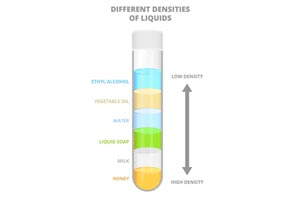
- Start your column by pouring the honey into the cylinder.
- Now, you will pour each liquid SLOWLY into the container, one at a time. It is better to pour the liquids slowly and into the very centre of the container. Try and make sure that the liquids do not touch the sides of the cylinder while you are pouring.
- It’s okay if the liquids mix as you are pouring in fact it looks cooler to see them separate out. The layers will always even themselves out because of the varying densities. After letting the liquid layers settle, you’ll notice that they remain in the order you poured them into the cylinder and that they are clearly distinguishable from each other.
- Make a chart that shows the order of each layer.
- Take the various small objects and drop them into the column. Metal nut or bolt, popcorn kernel, board game die, grape tomato, plastic bead, soda cap, ping pong ball. Do they float at different levels, if so, what does this mean?
How it works:
All materials, whether they are solid or liquid, have different densities and therefore will float at different levels.
Density is basically how much “stuff” is smashed into a particular area… or a comparison between an objects mass and volume. Remember the all-important equation: Density = Mass divided by Volume. Based on this equation, if the weight (or mass) of something increases but the volume stays the same, the density must go up. Likewise, if the mass decreases but the volume stays the same, the density must go down. Lighter liquids (like water or rubbing alcohol) are less dense than heavy liquids (like honey or syrup) and so float on top of the denser layers. The same goes for the small objects that you dropped into your density column. The metal bolt is denser than any of the liquids in the column and therefore sinks directly to the bottom. Less dense objects will float on individual layers of the column. However, the plastic bead is denser than the vegetable oil and everything above it but less dense than the water and everything below it. This makes the bead settle on the top of the water yet sink in the oil.





